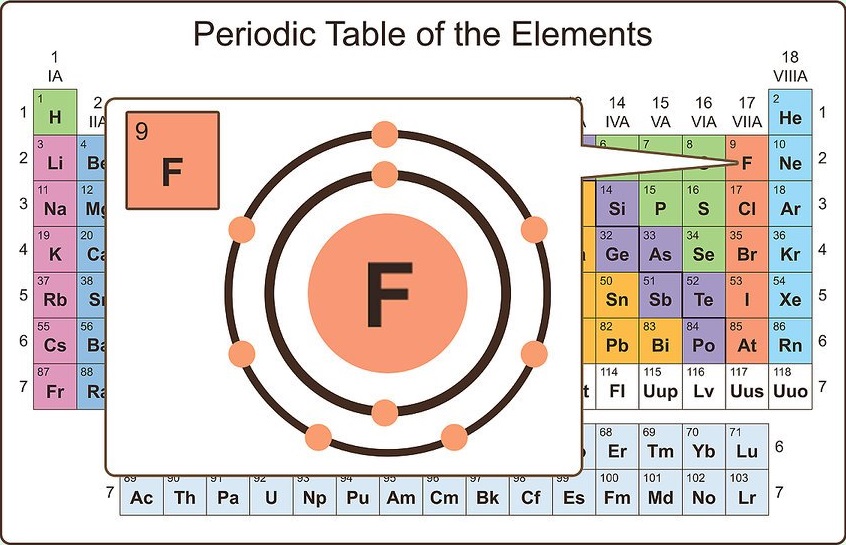
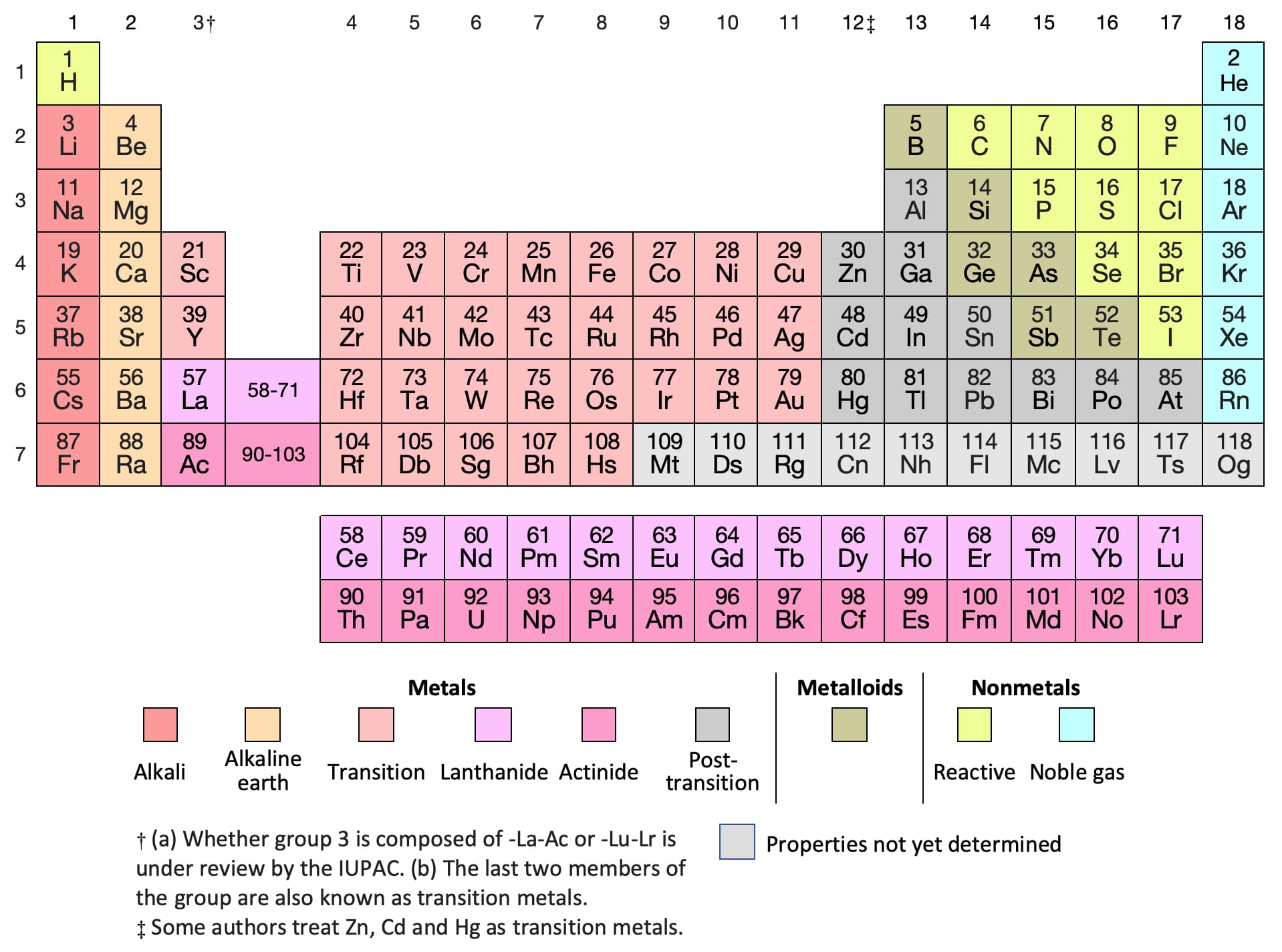
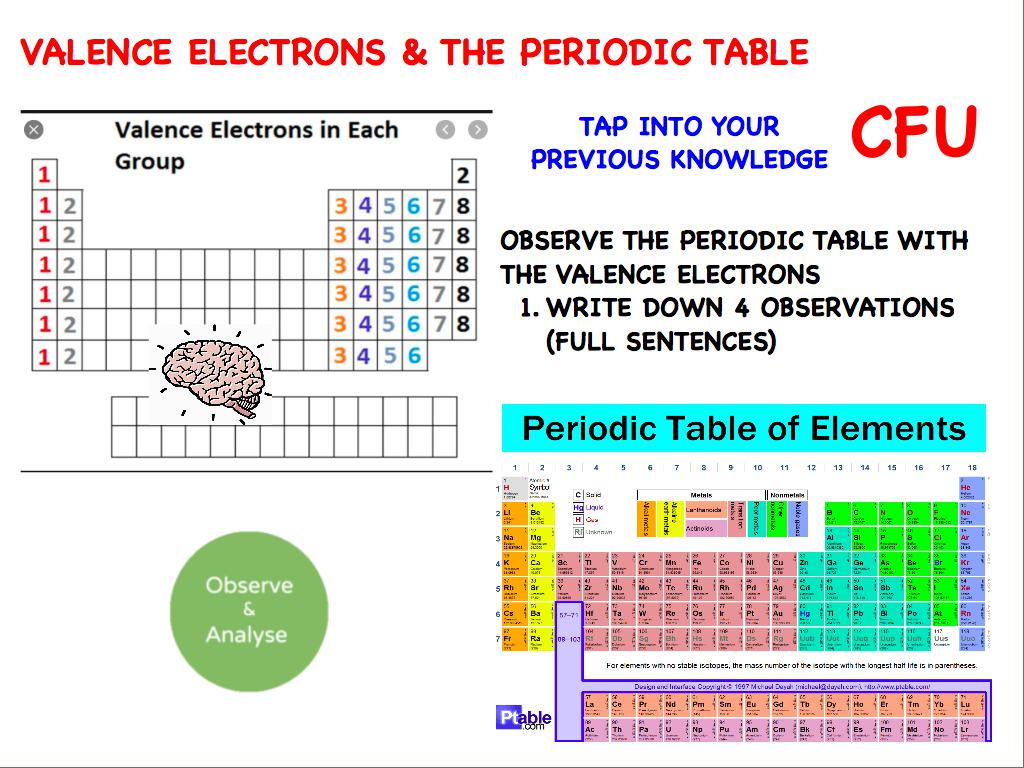
The number of valence electrons depends on the octet rule. Elements within the same group share the same number of valence electrons. Atoms in a period have the same number of electron shells. Of course, this is somewhat redundant, since the symbol Mg always implies Z=12, but it is sometimes a convenience when discussing several nuclides. An element period is a horizontal row on the periodic table. 1, reacts readily with metals to form compounds that can be broadly classified as salts and, therefore, are known as the halogens, which is derived from a combination of Greek words that translate to 'salt makers. A more explicit way of denoting a particular kind of nucleus is to add the atomic number as a subscript. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. Each of the elements found in this column, which are boxed in lavender in Figure 2.2.1 2.2. The element symbol depends on Z alone, so the symbol 26Mg is used to specify the mass-26 nuclide of magnesium, whose name implies Z=12. Remember, shells don’t neatly stack on top of each other, so valence (and oxidation state. The most stable oxidation state is one that fills or half-fills an atom’s electron shell. Usually it’s oxidation state is +1, but it can lose the electron and have a valence of -1. Any nuclide is characterized by the pair of numbers ( Z ,A). A lithium atom has one outer shell electron. For example, a nucleus of atomic number 7 is a nuclide of nitrogen. The term nuclide simply refers to any particular kind of nucleus. The chemical name of the element and its symbol are uniquely tied to the atomic number thus the symbol "Sr" stands for strontium, whose atoms all have Z = 38. You can think of the atomic number as a kind of serial number of an element, commencing at 1 for hydrogen and increasing by one for each successive element. Moseley found that the square roots of these frequencies give a straight line when plotted against Z this enabled him to sort the elements in order of increasing atomic number. The frequencies of these X-rays are unique to each element, and they increase uniformly in successive elements. He found this in a class of X-rays emitted by an element when it is bombarded with electrons. We can see from the electron configuration of a carbon atom1 s2 2 s2 2 p2 that it has 4 valence electrons (2 s2 2 p2) and 2 core electrons (1 s2 ). Moseley searched for a measurable property of each element that increases linearly with atomic number. Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. In the neutral atom, the Z protons within the nucleus are balanced by Z electrons outside it.Ītomic numbers were first worked out in 1913 by Henry Moseley, a young member of Rutherford's research group in Manchester. Each proton carries an electric charge of +1, so the atomic number also specifies the electric charge of the nucleus. It is, rather, the number of protons in the nucleus, which we call the atomic number and denote by the symbol Z. What single parameter uniquely characterizes the atom of a given element? It is not the atom's relative mass, as we will see in the section on isotopes below. The radius of an atom must be defined arbitrarily, such as the boundary in which the electron can be found with 95% probability. The latter has no definite outer boundary, so neither does the atom. It is common (but somewhat misleading) to describe the volume of space in which the electrons of an atom have a significant probability of being found as the electron cloud. The extremely small mass of the electron (1/1840 the mass of the hydrogen nucleus) causes it to behave as a quantum particle, which means that its location at any moment cannot be specified the best we can do is describe its behavior in terms of the probability of its manifesting itself at any point in space.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
